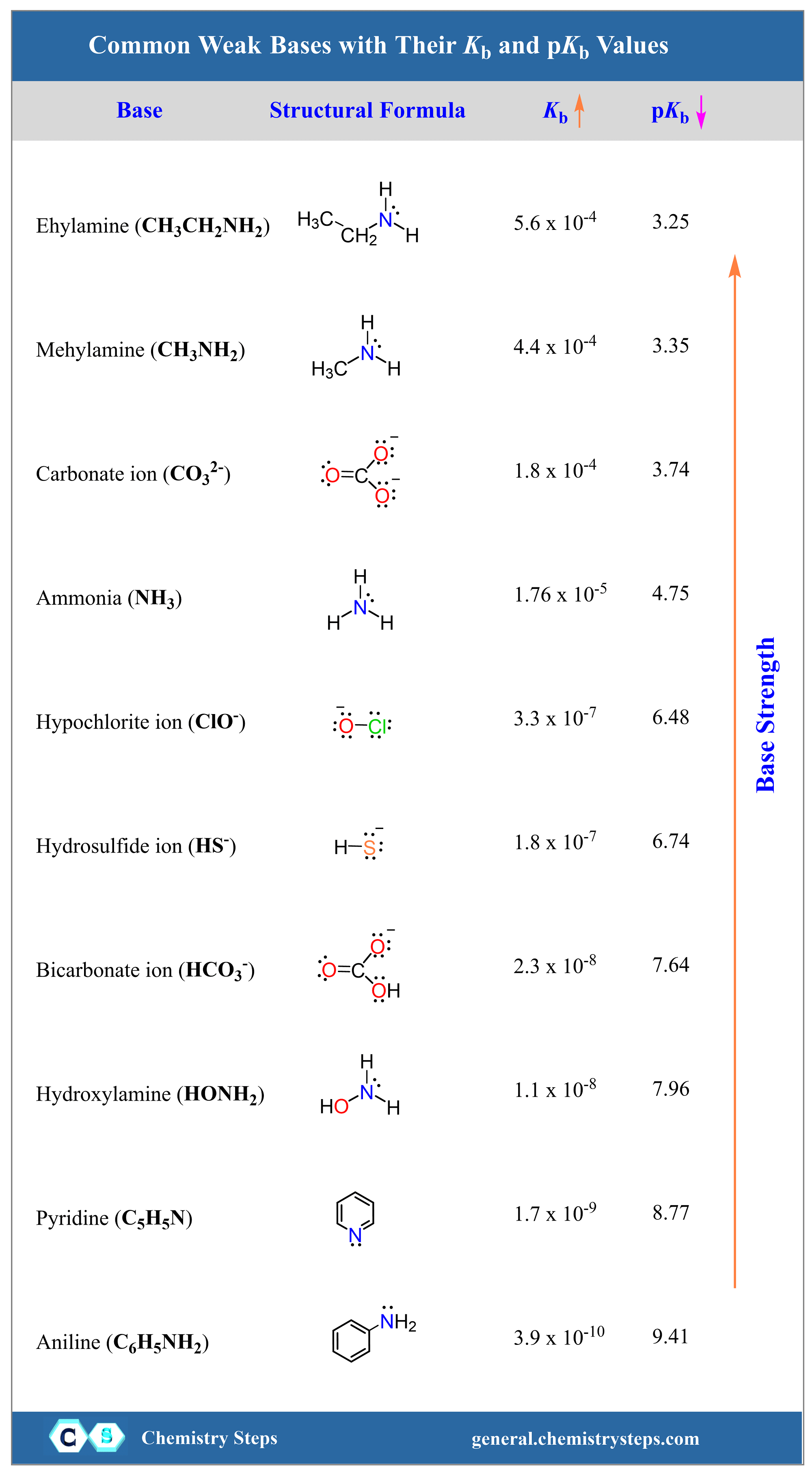
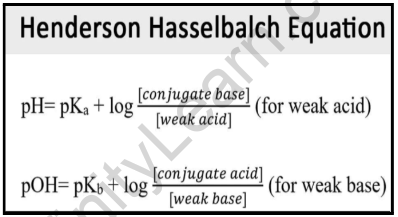
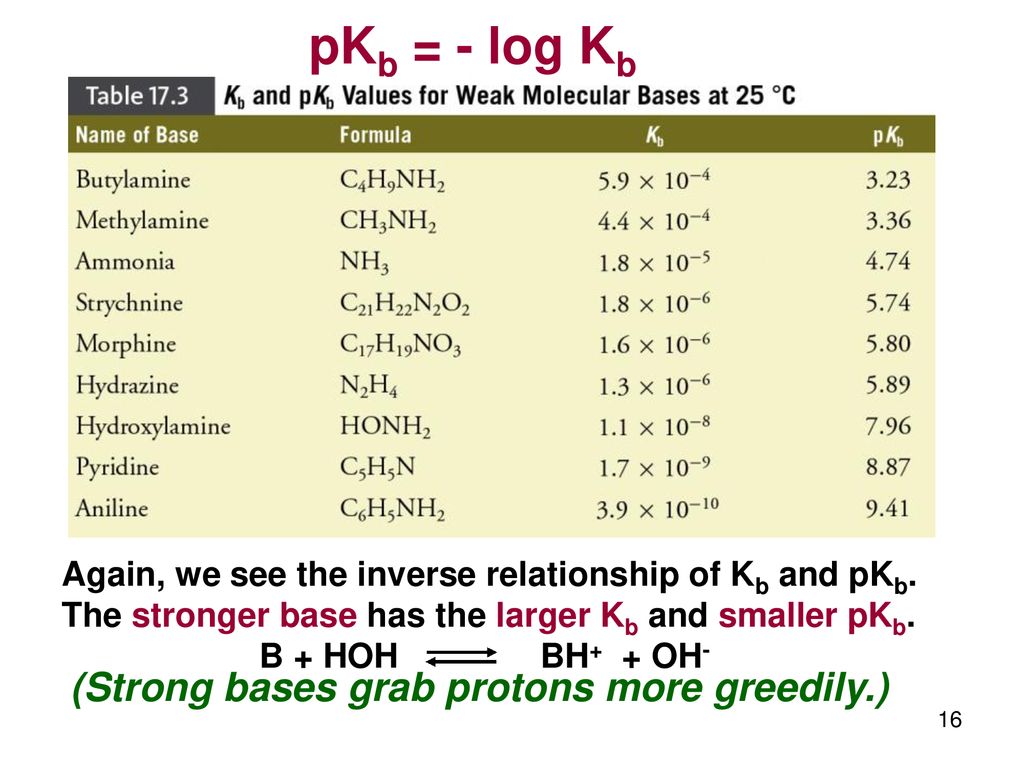
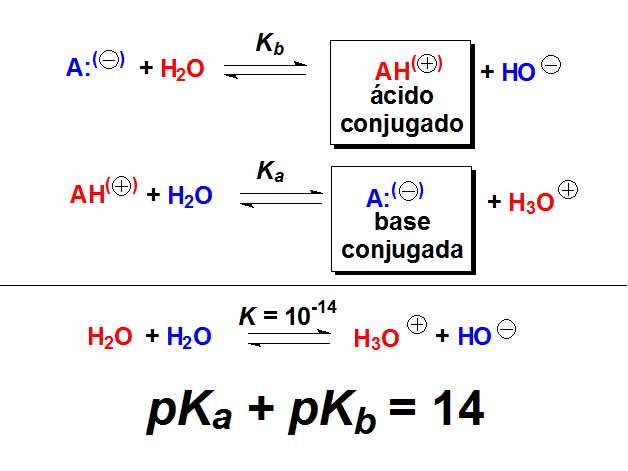
pKa of a weak acid (HA) and pKb of a weak base (BOH) are 3.2 and 3.4 respectively. The pH of their salt (AB) solution is? a) 7.2 b) 6.9 c) 7.0 d) 1.0
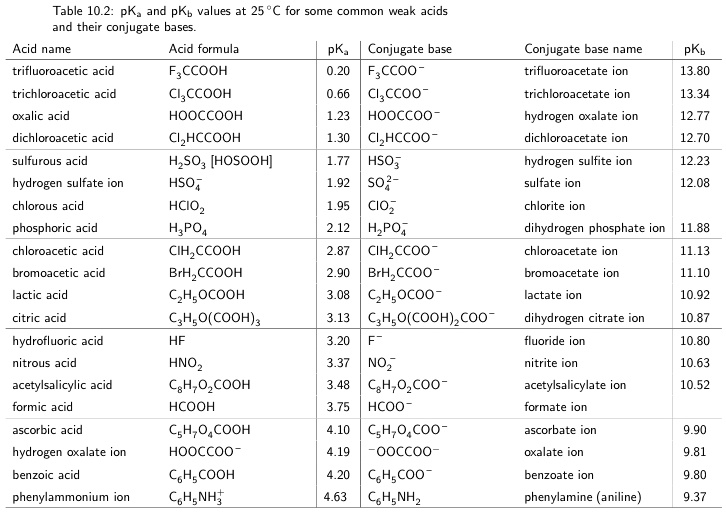
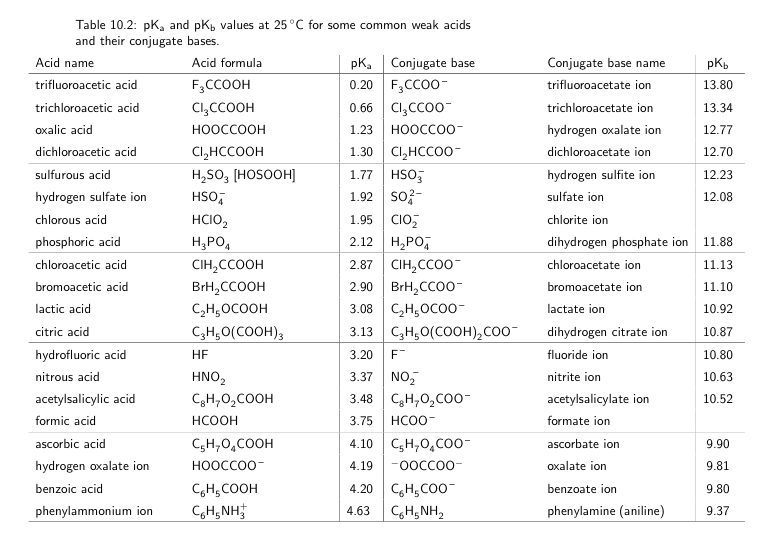
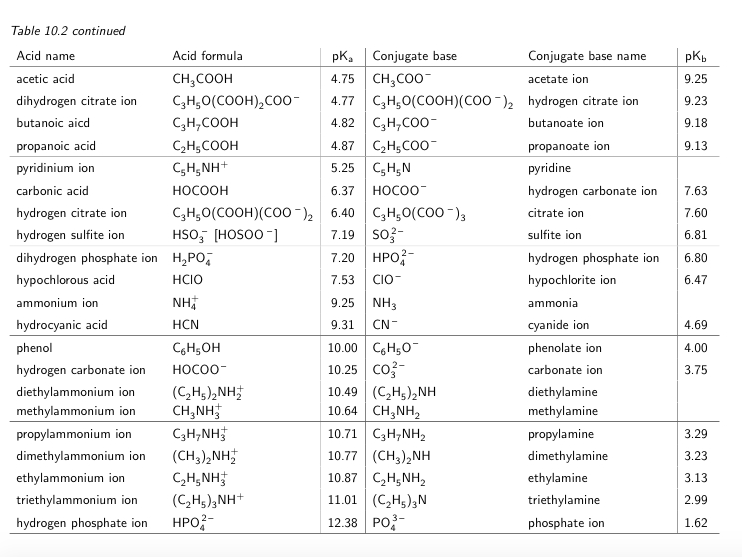
SOLVED: Table 10.2: pK pKb values at 25 C for some common weak acids and their conjugate bases: Acid name Acid formula pK Conjugate base trifluoroacetic acid FyCCooH FsCCOo trichloroacetic acid Clyccooh
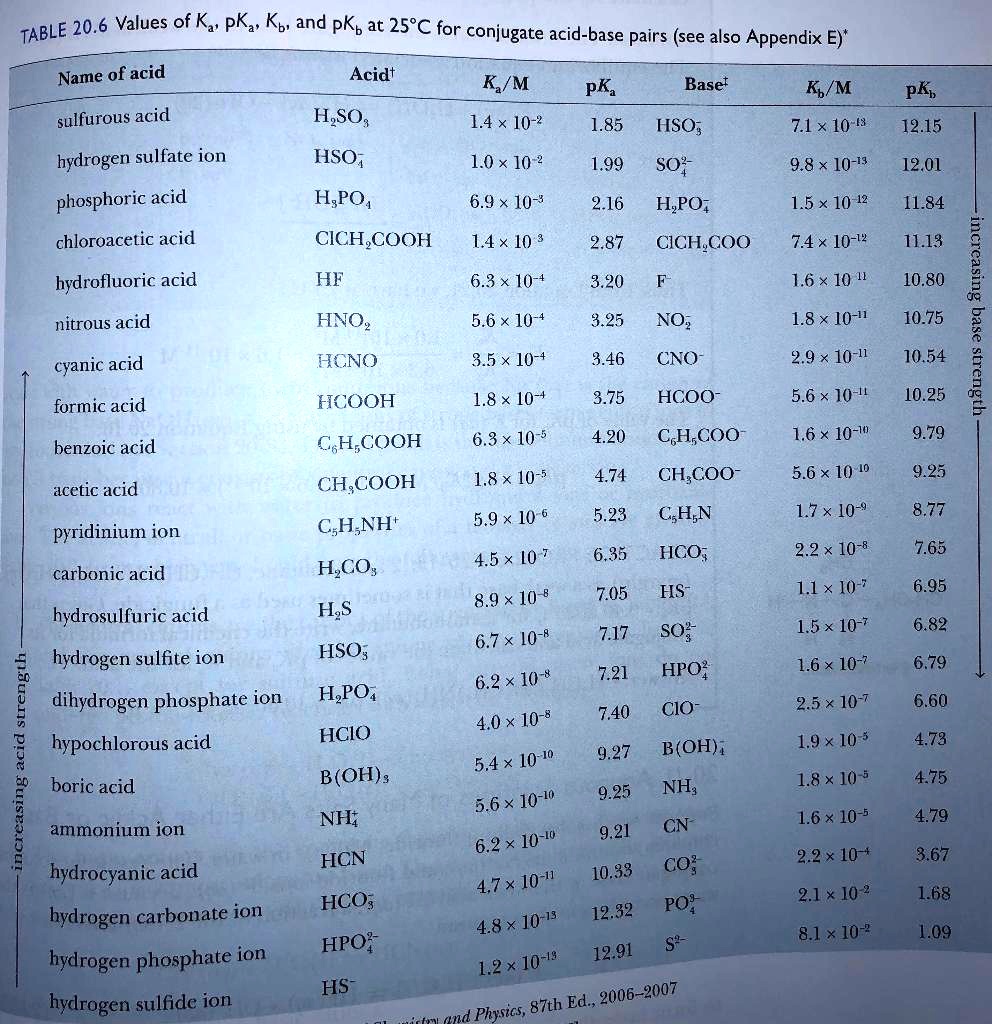
![The pKb of weak base BOH [Kb( BOH) = 1 xx 10^-5 ] will be The pKb of weak base BOH [Kb( BOH) = 1 xx 10^-5 ] will be](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/644557107_web.png)